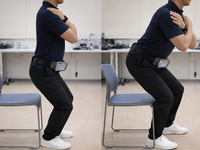
Validity of Smartphone-Based Measurement for Assessing Lower Limb Power for Sarcopenia and Frailty Discrimination: Cross-Sectional Study


Background: Men face a substantially higher risk of suicide. Effective suicide prevention strategies for men should specifically target gender-related risk factors, such as their lower likelihood of seeking professional help. Objective: This study investigates the use and impact of a suicide prevention website for men between March 1, 2023, and December 31, 2024. The Männer Stärken website is the first suicide prevention platform for men in Germany, with the primary aim of facilitating help-seeking behavior. The development of the platform was informed by interviews with men who had attempted suicide, as well as by existing evidence on effective communication strategies for engaging men at risk. Methods: This exploratory study combines quantitative web analytics and survey data with a qualitative analysis of open-ended responses from a feedback form. Using the web analytics tool Matomo, data were collected on the number of visits to the website and the subpages they accessed. In addition, 291 anonymous feedback forms were analyzed regarding visitors’ perceptions of the website’s helpfulness and its potential to support help-seeking behavior. A further component involved an online survey (n=40) examining whether a short suicide prevention film featured on the website could increase the intention to seek help. Results: During the study period, the website recorded 29,279 visits. A majority (n=291) of the respondents reported via the feedback form that they found the website helpful (n=201, 69.1%) and believed it could encourage help-seeking behavior (59.8%). In the evaluation of the short film, a significant increase in participants’ intentions to seek help was observed in situations involving suicidal ideation and personal difficulties and when considering professional support services. This effect was not observed with regard to informal sources of support, such as friends or family. Conclusions: The data suggest that the website is being used. Among those who completed the anonymous survey form (N=291), a majority reported that the website fulfills its primary aim of providing helpful pathways to support services. The evaluation of the short film further supports this conclusion. However, certain limitations must be acknowledged: since the data were collected in a field setting, the ability to draw firm conclusions about the characteristics or representativeness of the visitor sample is limited. In addition, the sample size for the short film evaluation was small. Nevertheless, the findings point to a clear need for gender-specific suicide prevention initiatives. They indicate promising directions for the development of effective, low-threshold measures, which merit further investigation in future research.
<img src="https://jmir-production.s3.us-east-2.amazonaws.com/thumbs/075608c52d64fbf781c284052cabc6f6" />
This story originally appeared in The Algorithm, our weekly newsletter on AI. To get stories like this in your inbox first, sign up here.
Within Silicon Valley’s orbit, an AI-fueled jobs apocalypse is spoken about as a given. The mood is so grim that a societal impacts researcher at Anthropic, responding Wednesday to a call for more optimistic visions of AI’s future, said there might be a recession in the near term and a “breakdown of the early-career ladder.” Her less-measured colleague Dario Amodei, the company’s CEO, has called AI “a general labor substitute for humans” that could do all jobs in less than five years. And those ideas are not just coming from Anthropic, of course.
These conversations have unsurprisingly left many workers in a panic (and are probably contributing to support for efforts to entirely pause the construction of data centers, some of which gained steam last week). The panic isn’t being helped by lawmakers, none of whom have articulated a coherent plan for what comes next.
Even economists who have cautioned that AI has not yet cut jobs and may not result in a cliff ahead are coming around to the idea that it could have a unique and unprecedented impact on how we work.
Alex Imas, based at the University of Chicago, is one of those economists. He shared two things with me when we spoke on Friday morning: a blunt assessment that our tools for predicting what this will look like are pretty abysmal, and a “call to arms” for economists to start collecting the one type of data that could make a plan to address AI in the workforce possible at all.
On our abysmal tools: consider the fact that any job is made up of individual tasks. One part of a real estate agent’s job, for example, is to ask clients what sort of property they want to buy. The US government chronicled thousands of these tasks in a massive catalogue first launched in 1998 and updated regularly since then. This was the data that researchers at OpenAI used in December to judge how “exposed” a job is to AI (they found a real estate agent to be 28% exposed, for example). Then in February, Anthropic used this data in its analysis of millions of Claude conversations to see which tasks people are actually using its AI to complete and where the two lists overlapped.
But knowing the AI exposure of tasks leads to an illusory understanding of how much a given job is at risk, Imas says. “Exposure alone is a completely meaningless tool for predicting displacement,” he told me.
Sure, it is illustrative in the gloomiest case—for a job in which literally every task could be done by AI with no human direction. If it costs less for an AI model to do all those tasks than what you’re paid—which is not a given, since reasoning models and agentic AI can rack up quite a bill—and it can do them well, the job likely disappears, Imas says. This is the oft-mentioned case of the elevator operator from decades ago; maybe today’s parallel is a customer service agent solely doing phone call triage.
But for the vast majority of jobs, the case is not so simple. And the specifics matter, too: Some jobs are likely to have dark days ahead, but knowing how and when this will play out is hard to answer when only looking at exposure.
Take writing code, for example. Someone who builds premium dating apps, let’s say, might use AI coding tools to create in one day what used to take three days. That means the worker is more productive. The worker’s employer, spending the same amount of money, can now get more output. So then will the employer want more employees or fewer?
This is the question that Imas says should keep any policymaker up at night, because the answer will change depending on the industry. And we are operating in the dark.
In this coder’s case, these efficiencies make it possible for dating apps to lower prices. (A skeptic might expect companies to simply pocket the gains, but in a competitive market, they risk being undercut if they do.) These lower prices will always drive some increase in demand for the apps. But how much? If millions more people want it, the company might grow and ultimately hire more engineers to meet this demand. But if demand barely ticks up—maybe the people who don’t use premium dating apps still won’t want them even at a lower price—fewer coders are needed, and layoffs will happen.
Repeat this hypothetical across every job with tasks that AI can do, and you have the most pressing economic question of our time: the specifics of price elasticity, or how much demand for something changes when its price changes. And this is the second part of what Imas emphasized last week: We don’t currently have this data across the economy. But we could.
We do have the numbers for grocery items like cereal and milk, Imas says, because the University of Chicago partners with supermarkets to get data from their price scanners. But we don’t have such figures for tutors or web developers or dietitians (all jobs found to have “exposure” to AI, by the way). Or at least not in a way that’s been widely compiled or made accessible to researchers; sometimes it’s scattered across private companies or consultancies.
“We need, like, a Manhattan Project to collect this,” Imas says. And we don’t need it just for jobs that could obviously be affected by AI now: “Fields that are not exposed now will become exposed in the future, so you just want to track these statistics across the entire economy.”
Getting all this information would take time and money, but Imas makes the case that it’s worth it; it would give economists the first realistic look at how our AI-enabled future could unfold and give policymakers a shot at making a plan for it.
OneCyte, which focuses on high-throughput single-cell analysis and cell line development technologies, and Kemp Proteins, which specializes in protein engineering and expression solutions, signed a strategic partnership agreement to deliver cell line development services for biopharmaceutical companies.
The collaboration brings together OneCyte’s proprietary single-cell platform for high-throughput and high-speed clone selection with Kemp Proteins’ molecular engineering capabilities, including its machine learning–driven platform, PROTiQ .
.
Biopharma companies continue to face significant challenges in cell line development, including long development cycles, suboptimal yields, and high failure rates, particularly for novel and complex molecules, according to Konstantinos Tsioris, PhD, co-founder and president of OneCyte. These challenges can delay regulatory timelines and slow the progression of therapies into the clinic.
The OneCyte-Kemp partnership addresses these pain points by integrating predictive in silico design with rapid and high throughput experimental validation, say officials at both companies. As part of the workflow, amino acid sequences are evaluated using Kemp’s PROTiQ platform to assess developability risks, identify sequence liabilities, and generate structural insights.
The optimized candidates are then paired with OneCyte’s high-performance cell line development platform, which reportedly enables identification of elite clones with higher productivity.
Unlike traditional, rigid development workflows, this integrated approach is designed to adapt quickly to the evolving needs of new therapeutic modalities, notes Tsioris.
“By combining our single-cell technology with Kemp’s deep expertise in protein expression, we are confident that we can address the hardest challenges associated with new modalities, delivering faster timelines and industry-leading titers,” he continues.
“OneCyte’s class-leading single-cell technology, stacked on top of our molecular design and expression capabilities, will provide a powerful and differentiated solution for our global biopharma customers,” says Michael Keefe, CEO of Kemp Proteins.
The post Machine Learning and Single-Cell Technology Combined to Drive High-Performance Cell Line Development appeared first on GEN – Genetic Engineering and Biotechnology News.
This is the online version of STAT’s weekly email newsletter Health Care Inc. Sign up here.
Well hullo! You think you can get the last word, literally, with AI? Think again. There’s always human interaction available here: bob.herman@statnews.com.
By law, the 2027 Medicare Advantage payment regulation must come out today. It will set the tone for how the Trump administration wants to work with the health insurance industry: as the “new sheriff in town” or just another friendly regulator.
Central nervous system (CNS) treatments are having a major comeback. These five precision medicine players plan to ride the resurgence.
After a decade of stagnation, the CNS space is seeing a revival in sales and R&D spending as the market was last year projected to surpass $80 billion for the first time since 2013 and hit around $127 billion.
Recent landmark approvals have brought attention back to the CNS, including the U.S. Food and Drug Administration (FDA)’s greenlight of Eisai/Biogen’s lecanemab (Leqembi) for the treatment of Alzheimer’s disease in 2023, and the FDA approval of Bristol-Myers Squibb’s schizophrenia treatment xanomeline/trospium chloride (Cobenfy) in 2024.
At the same time, Johnson & Johnson’s depression treatment, esketamine (Spravato), is on its way to blockbuster status, showcasing the growth potential of the CNS market.
These successes accompany an emerging shift in psychiatry clinical trials from subjective rating scales to more objective endpoints, including digital and physiological measures, with the potential to better tailor treatments to a patient’s biological makeup.
Startups and scaleups are attracting increasing investor attention for their potential to change the way we treat CNS conditions. Check out our list of the most exciting companies that have netted the biggest investor dollars.
Founded: 2025 | Headquarters: Dublin, Ireland
Aerska’s name is derived from an Irish proverb stating that people survive in each other’s shelter, emphasising the strength of its team.
This team includes co-founder Jack O’Meara, previously co-founder of the liver-focused RNA interference (RNAi) biotech Ochre Bio, who is driven by the experience of loved ones suffering from Alzheimer’s disease.
Aerska is developing RNAi therapies for neurodegenerative conditions, including Parkinson’s and Alzheimer’s disease.
While there are already FDA-approved RNAi therapies on the market, such as Alnylam’s patisiran (Onpattro), these are typically focused on liver and cardiometabolic conditions rather than the CNS.
Aerska’s technology consists of antibody “brain shuttles” that bind to proteins on the blood-brain barrier (BBB). They then carry a payload RNA into the brain.
The payload, which is designed based on data-driven patient stratification and disease biomarkers, then silences specific genes driving the disease.
Aerska has already raised $60 million since its launch, including a $21 million seed round in October 2025 and a $39 million Series A round in February 2026, co-led by EQT Life Sciences and age1.
The company, which has research operations in the U.K., is using the latest funding to drive its pipeline programs toward clinical testing.
Founded: 2019 | Headquarters: Boston, Massachusetts, U.S.
Beacon Biosignals was co-founded by a team including its CEO—MIT neuroscientist Jacob Donoghue, MD, PhD—and its CTO, the machine learning researcher Jarrett Revels.
Boasting more than 100 employees, the company’s goal is to provide objective biomarkers in drug development that neurology and psychiatry have traditionally lacked compared with other areas of precision medicine.
Its FDA-cleared Waveband device measures the brain’s activity, known as electroencephalography (EEG), while patients sleep at home. The EEG data is then stored, quality-controlled, and fed into AI models that can guide the design of clinical trials.
For example, Beacon’s EEG data can identify patients with Alzheimer’s disease who have worse outcomes and might need a more targeted treatment or a different clinical trial than other patients.
Beacon raised $27 million in a Series A round in 2021 and an oversubscribed Series B round worth $86 million in November 2025.
The B round, which included investors such as Innoviva, Google Ventures, and Nexus NeuroTech, will help the startup to accelerate the discovery of neurobiomarkers and broaden clinical adoption of the technology.
Beacon acquired the French sleep monitoring company Dreem in 2023 to access its monitoring data and headband technology. Beacon then acquired the Ohio-based CleveMed in April 2025 to harness technology measuring breathing, oxygen, and other signals.
Founded: 2010 | Headquarters: Oxford, U.K.
Brainomix was founded by a team including CEO Michalis Papadakis, PhD, who was scientific director of the preclinical stroke lab at the University of Oxford.
Brainomix is dedicated to speeding up patient care in cases of stroke, where speedy treatment is key.
Brainomix’s flagship product, Brainomix 360 Stroke, is designed to harness AI to interpret brain scans and detect blood clots in patients with stroke, speeding up clinical decision-making.
The product involves a group of tools that automatically analyze images, including results from computed tomography (CT), CT angiography, magnetic resonance imaging (MRI), and CT perfusion.
Brainomix’s technology doubled the rate of thrombectomy treatment in patients with stroke and reduced hospital triage and transfer delays, according to a 2025 study.
The University of Oxford spinout is at a commercial stage, with operations in more than 20 countries, and is expanding into the U.S.
Brainomix raised a $21.2 million Series B round in 2021 and extended its Series C round from $6.5 million in March 2025 to $25.4 million in February 2026, with leading investors including Parkwalk Advisors and Hostplus. The proceeds will fuel the company’s expansion into the U.S. market.
Brainomix has also partnered with heavyweights, including Nvidia, Boehringer Ingelheim, Medtronic, and GE Healthcare.
Brainomix also has a product dedicated to disease monitoring in pulmonary fibrosis.
Founded: 2021 | Headquarters: San Diego, California, U.S.
Circular Genomics was spun out of the University of New Mexico, with its founders including CSO Nikolaos Mellios, PhD, and Alexander Hafez, PhD.
The company later moved its headquarters from Albuquerque to San Diego in March 2025 to access scientific and operational know-how from Eli Lilly at Lilly Gateway Labs.
Circular Genomics aims to equip medical professionals with a blood test to detect CNS conditions early, in addition to stratifying and guiding the treatment of patients.
Its technology involves using a polymerase chain reaction (PCR) test of a patient’s blood sample to screen for specific circular RNA molecules produced in the brain that can cross into the blood and be measured as a biomarker of disease in the CNS.
Commercially launched in 2024, Circular Genomics’ MindLight SSRI Antidepressant Response Test predicts whether a patient will benefit from common antidepressants called SSRIs with around 77% accuracy. This is designed to predict a patient’s most suitable antidepressants without needing months of trial-and-error approaches.
The company is applying its technology in Alzheimer’s disease, where the approvals of disease-modifying therapies such as Leqembi have led to demand for tests that can detect the disease at earlier stages than traditional tests.
Circular Genomics raised $15 million in a Mountain Group Partners-led Series A round in December 2025 to finance the development of its technology and expansion of its technology in Alzheimer’s disease.
The company also has its sights on other CNS conditions, including multiple sclerosis and Parkinson’s disease.
Founded: 2019 | Headquarters: Sydney, Australia
Omniscient (o8t)’s founders include CMO Michael Sughrue, MD, a neurosurgeon aiming to improve anatomy maps for other surgeons, and machine learning expert Stephane Doyen, PhD.
o8t’s FDA-approved product Quicktome involves using a patient’s MRI brain scans and AI models to map out a patient’s brain circuitry. These maps, accessible from an electronic tablet, can guide surgery to minimize the risk of brain damage compared to using a generalized anatomical diagram.
Quicktome is already in use at major hospitals around the world, including major centers in the U.S. Its partners include U.S. surgical support firm META Dynamic and the U.S. medical device innovation center, The Jacobs Institute.
o8t has raised more than $60 million, and bagged $14 million (AUD 20 million) in January 2026 as part of a Series D round targeted to reach $25 million (AUD 36 million). The round was led by Australia’s National Reconstruction Fund (NRFC) and OIF Ventures, with the aim of keeping the company based in Australia.
The funding is earmarked to fuel the development and commercialization of Quicktome, and grow o8t’s Australian workforce by more than 40. The company also has operations in Atlanta, Georgia, U.S.
o8t also plans to expand the technology into high-growth markets, including brain computer interface targeting, stroke and traumatic brain injury.
Jonathan Smith, PhD, is a freelance science journalist based in the U.K. and Spain. He previously worked in Berlin as a reporter and news editor at Labiotech, a website covering the biotech industry. Prior to this, he completed a PhD in behavioral neurobiology at the University of Leicester and freelanced for the U.K. organizations Research Media and Society of Experimental Biology. He has also written for medwireNews, Biopharma Reporter, and Outsourcing Pharma.
The post Top 5 Firms Engineering Healthcare in the CNS Space appeared first on Inside Precision Medicine.
Good morning, everyone, and welcome to another working week. We hope the weekend respite was relaxing and invigorating because that oh-too-familiar routine of meetings, deadlines, and the like has returned with a vengeance. You knew this would happen, yes? To cope, we are relying, as always, on cups of stimulation. Our choice today is laced with traces of cocoa. Feel free to join us. Remember, no prescription is required. Meanwhile, here are some tidbits to help you along. Best of luck accomplishing your goals today and, of course, do keep in touch. …
In February, a small biotech company called Kezar Life Sciences reached a breakthrough with the U.S. Food and Drug Administration, agreeing to a plan for a clinical trial it hoped could lead to the approval of its treatment for a rare, debilitating liver disease called autoimmune hepatitis. The problem: The agreement came four months too late, STAT explains. The meeting to discuss trial design, a critical step in the drug development process, had been scheduled for last October. But the FDA abruptly canceled it without explanation. The company could no longer proceed as planned and, without clarity from regulators, its path forward was unclear. Kezar’s investors wanted out, and the biotech was forced to start the process of winding down.
Americans starting weight loss medicines for the first time want lower cost and greater convenience as they consider pills from Novo Nordisk and Eli Lilly, Reuters says, citing seven doctors who specialize in obesity. Novo’s Wegovy pill has been on the market since January, while Lilly’s newly approved Foundayo joins the fray this week. Interviews with the specialists show a promising landscape for oral weight loss drugs as the companies compete for share in the fast-changing obesity treatment market that is seen topping $100 billion a year in the next decade. All seven doctors said they had begun prescribing oral Wegovy, and three said they have prescribed the pill to about 10% of their patients. Of those patients, most are taking a GLP-1 for the first time, rather than switching from injectables, and have not yet reached the highest dose.